CMI researchers at Oak Ridge Laboratory and Purdue University conducted the activity for this highlight
Innovation
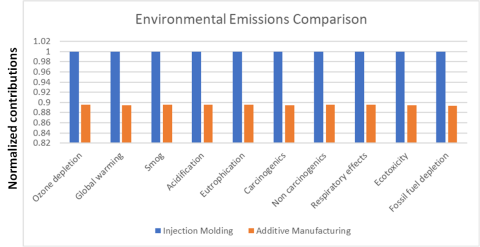
Additive Manufacturing (AM) or 3D printed NdFeB bonded magnets outperform conventional injection molded magnets at lower environmental impact.
Achievement
- AM methods demonstrated the gap magnet target with (BH)max of > 21 MGOe.
- Near-net-shape magnets were obtained without the need for complex injection molding tooling.
Significance and Impact
Big Area Additive Manufacturing (BAAM) printers at scale yield a significant decrease in critical rare earth mineral consumption and environmental emissions.
Hub Target Addressed
Develop methods to reduce the environmental impacts of rare earth magnet manufacturing.
