CMI researchers at Ames Laboratory conducted the research for this highlight
Achievement
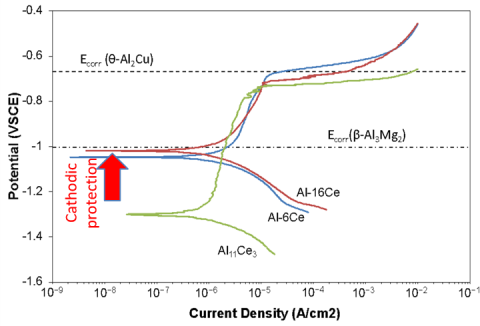
- Electrochemical properties of Al-Ce alloys the majority binary intermetallic show highly anodic potential compared to other phases common in Al alloys.
- Through targeted additions of Ce to A356 and other alloys it is shown that presence of Ce inhibits corrosive attack and reduces alloy degradation in harsh environments.
Significance and impact
- Cerium is a common material and can be added directly to melts across the aluminum industry to impart improved corrosion resistance.
- The benefits of Ce corrosion inhibition are not tied solely to the binary intermetallic and extend to higher order compositions. Creating the opportunity for compounding benefits with tailored compositional modification.