Overview
Separation science plays a vital role in the U.S. economy, with estimates that separation processes account for up to 15% of the total energy used. Ionic liquids (IL) and deep eutectic solvents (DES) are solvents that hold promise in reducing energy consumption when employed in separation systems. The team’s overarching hypothesis is that nanoscale spatial and chemical inhomogeneities are important to many aspects of molecular transport and partitioning in separation processes that use these solvents; furthermore, that these inhomogeneities can be tuned and modulated to improve separation performance. The existence of nanodomains, defined as nanometer-sized regions within the solvent formed by aggregation of charged ions and uncharged alkyl chains from the cation, is known in many ILs. However, there has been no attempt to correlate their presence, composition, and structure to separation performance.
The team proposes to uncover the nature of these inhomogeneities by employing a suite of unique chromatographic and optical techniques pioneered by the team and designed to measure nanoscale structure in complex systems. Experimental data will drive the development of theoretical models that will provide an understanding of how IL and DES structural features affect bulk and interfacial nanoscale ordering. The information harnessed by the team will stimulate the development of separations that are faster and require less energy input. The team is poised to succeed in this endeavor owing to its expertise in separation science and its access to expertise in employing methods and tools uniquely available at Ames Laboratory.
This research was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division, Separation Science program. Ames National Laboratory is operated for the U.S. Department of Energy by Iowa State University under Contract No. DEAC02-07CH11358.
Principal Investigator: Jared Anderson
Co-PIs: Emily Smith, Jake Petrich, Xueyu Song
News & Highlights
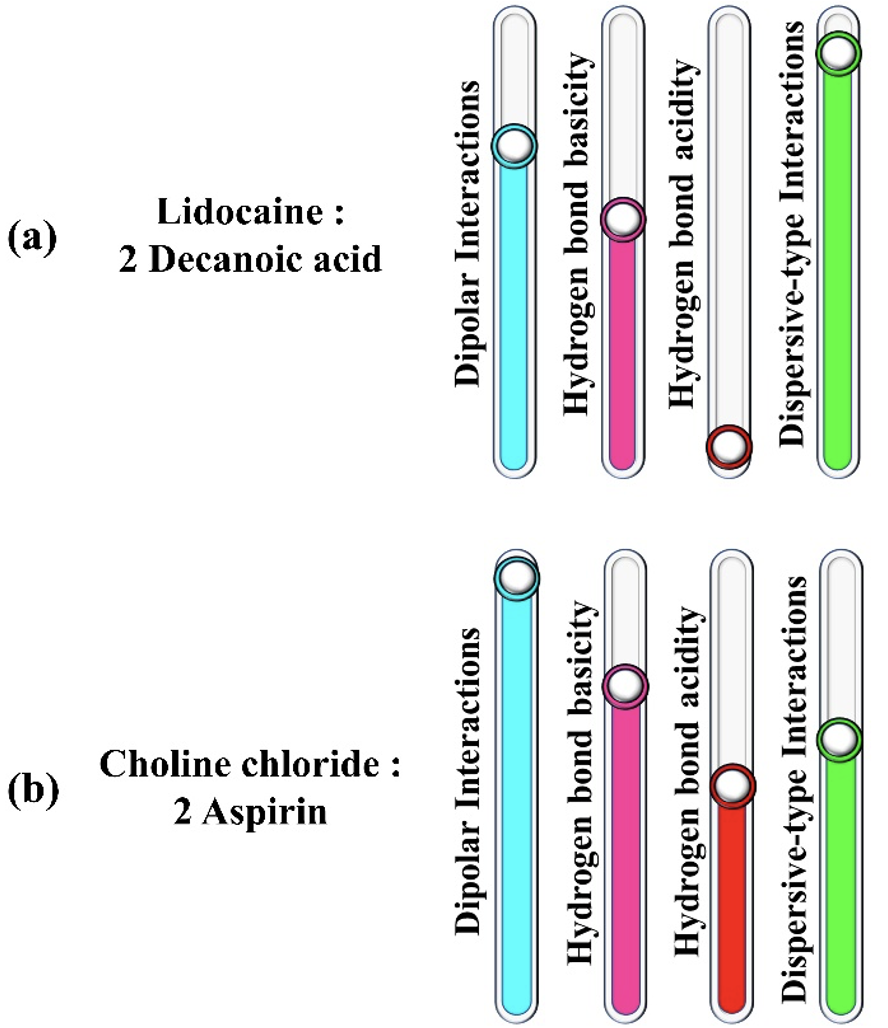 Research Highlight
Research Highlight
Characterizing the Solvation Characteristics of Deep Eutectic Solvents Composed of Active Pharmaceutical Ingredients
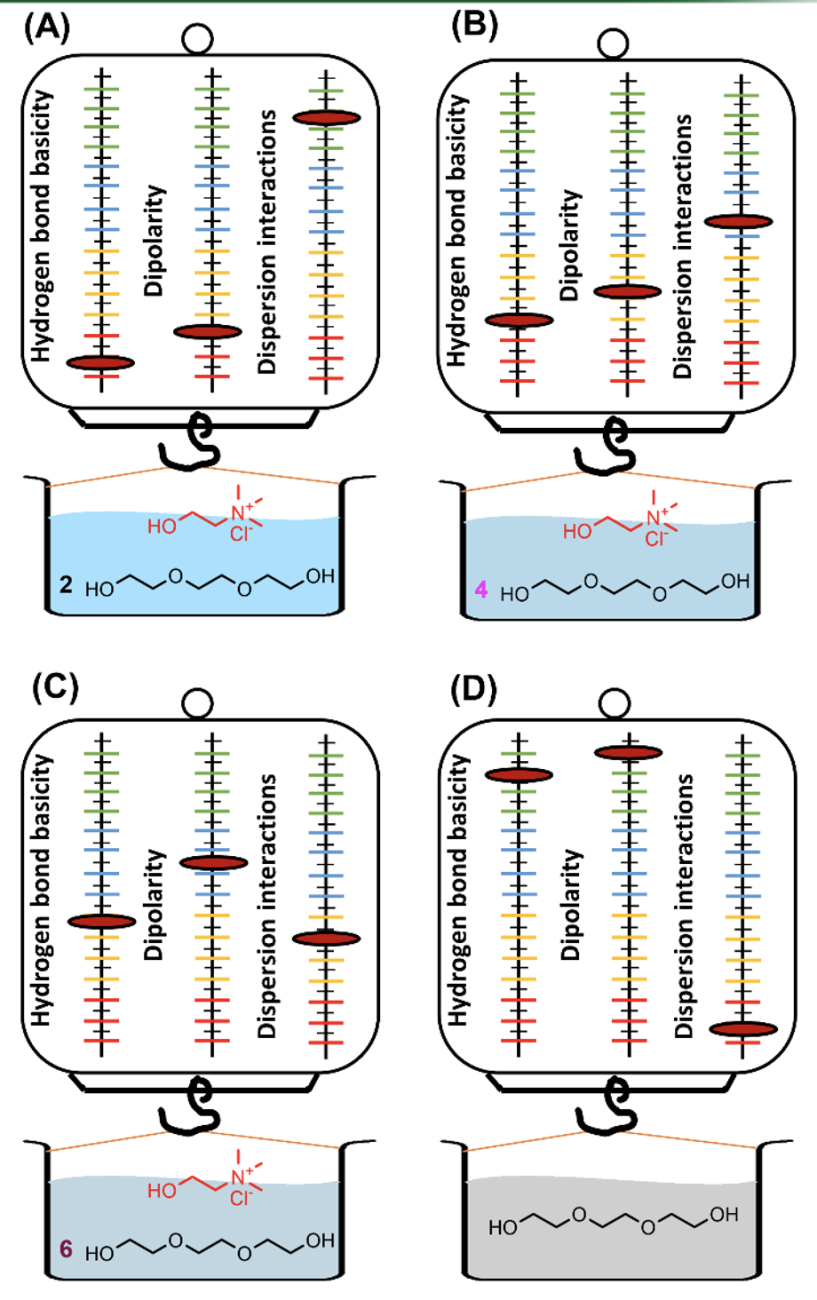 Research Highlight
Research Highlight
Investigating the Solvation Interactions of Deep Eutectic Solvents Formed Using Choline Chloride and Polyalcohols
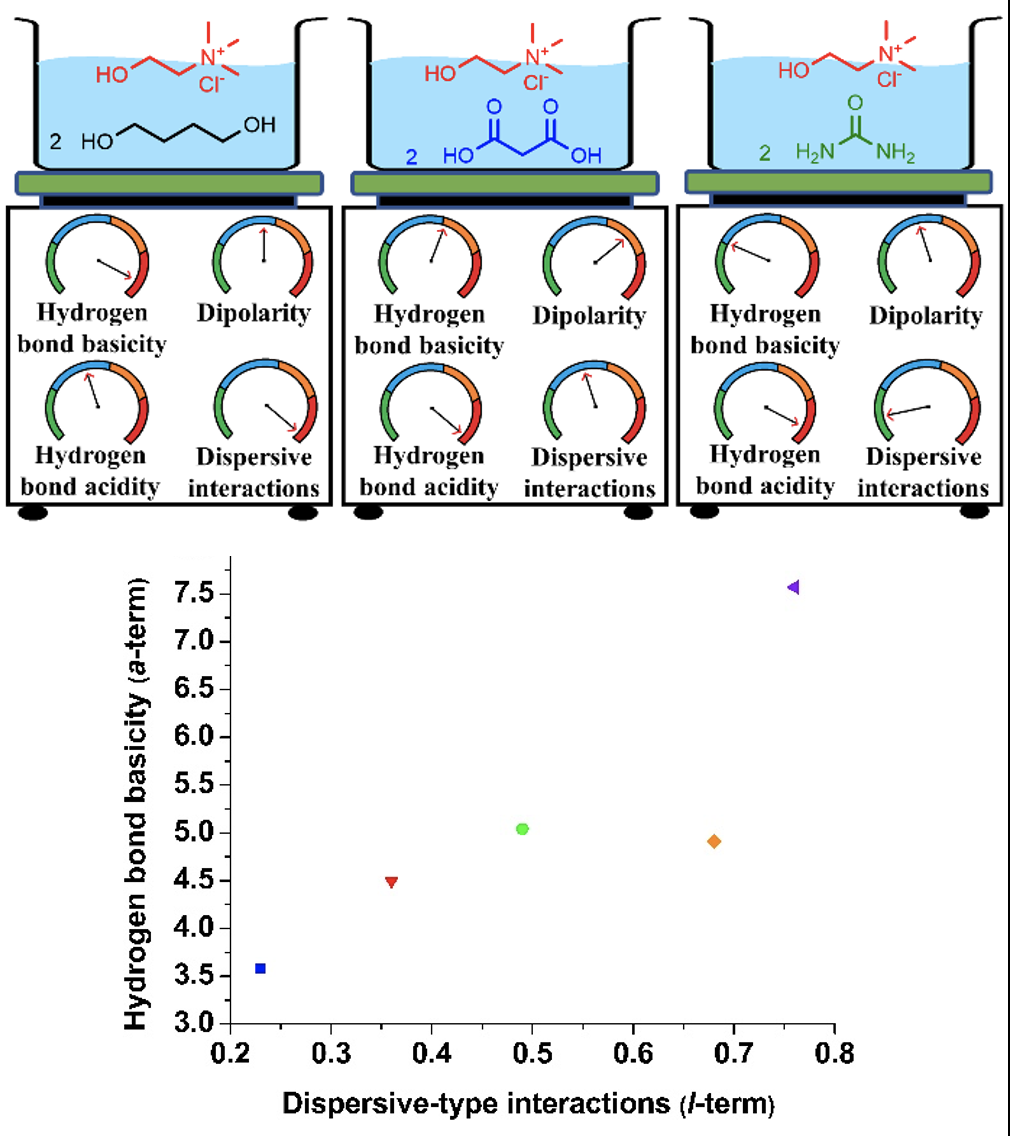 Research Highlight
Research Highlight
Characterizing the Solvation Interactions of Choline Chloride-based Deep Eutectic Solvents Employed in Separations
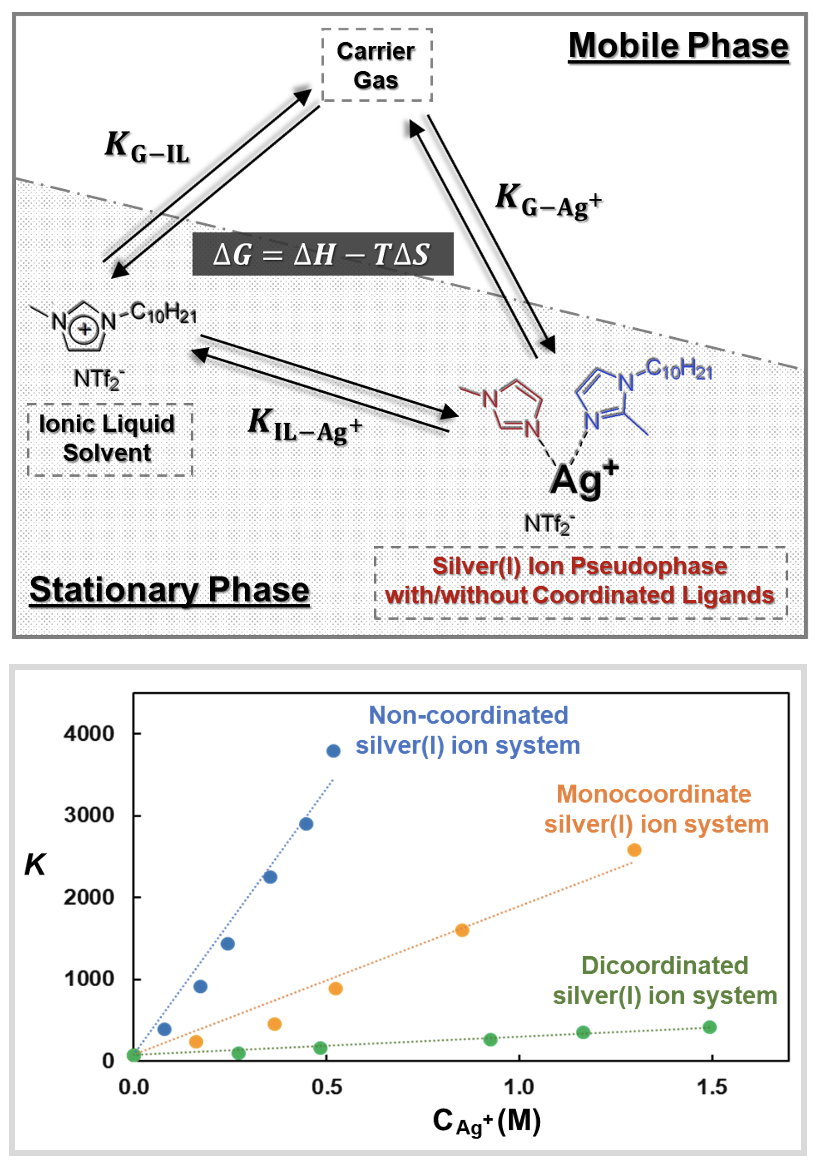 Research Highlight
Research Highlight
Using a Chromatographic Pseudophase Model to Elucidate the Mechanism of Olefin Separation by Silver(I) Ions in Ionic Liquids
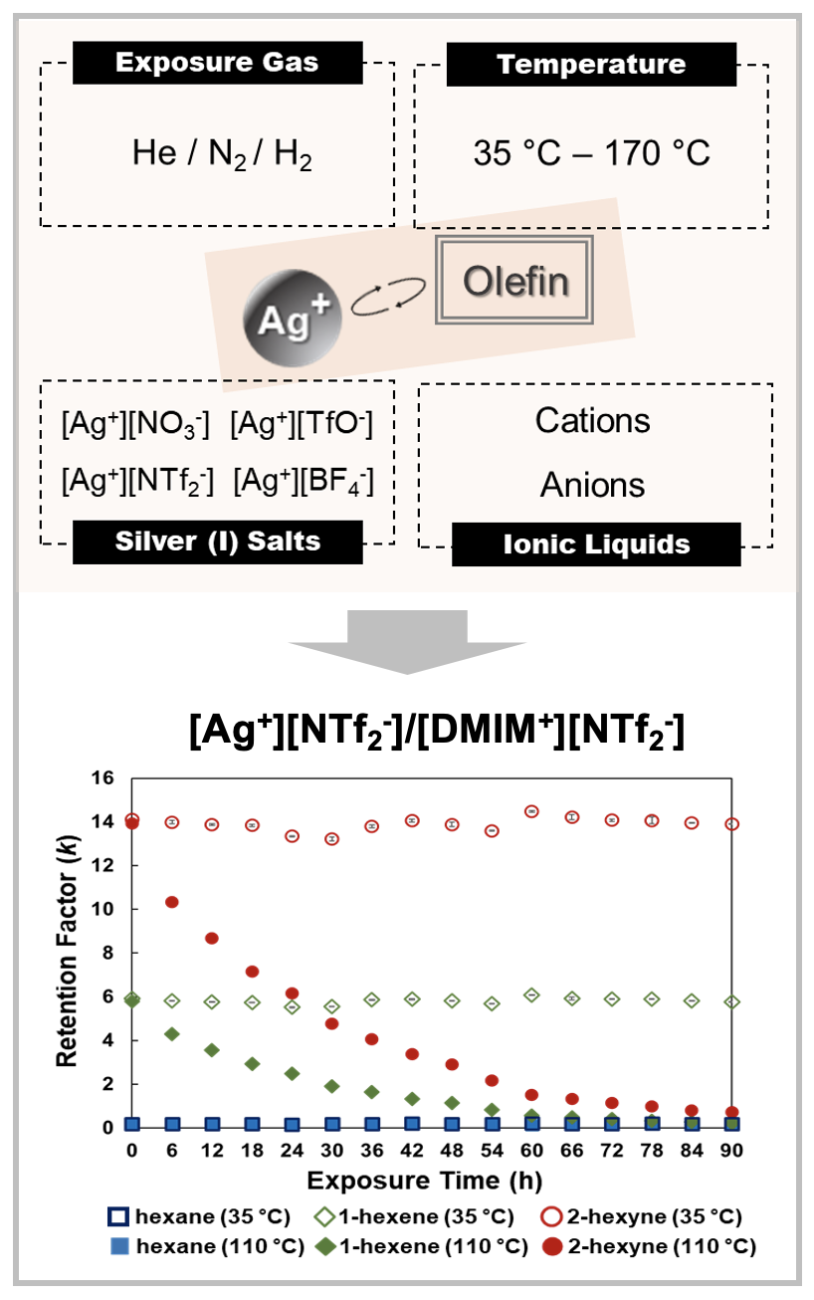 Research Highlight
Research Highlight
