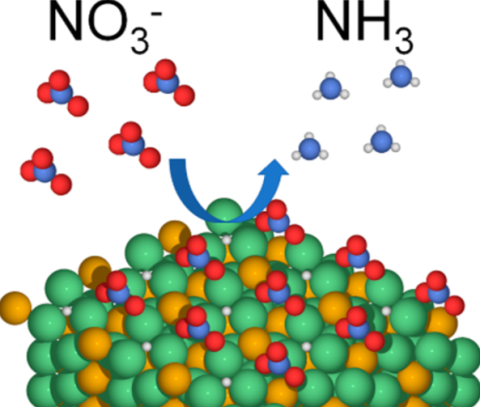
a Ni-rich Ni2P(001) surface exposed to hydrogen. NO3- must adsorb on the H-populated catalysis surface for hydrogenation (H: small white spheres; N blue; O: red; Ni: green; P: gold).
Access to clean water is an increasing challenge, particularly since higher demand for agricultural products incentivizes fertilizer use leading to stream contamination with nitrogen and phosphorous. Removing nitrate is difficult due to its thermodynamic stability, and is expensive using physical separation methods. Alternative chemical degradation methods typically exploit scarce and expensive precious metal catalysis. Given indications that binary phosphides are excellent candidates as catalysts for the hydrogen evolution reaction, we considered Ni2P as a candidate for nitrate hydrogenation. This catalyst proved to be both effective and also selective to ammonia production.
Plane-wave DFT analysis using slab geometries has proven a powerful tool for detailed analysis of heterogeneous catalysis on single-crystal surfaces. DFT is applied here first to characterize different reconstructions of the Ni2P surface, and then to analyze H adsorption characteristics thought to control catalysis effectiveness. Elucidation of this effectiveness is aided by comparison with behavior on traditional fcc(111) metal catalysts surfaces.
Various reconstructed and unreconstructed surface of Ni2P provide a network of 3 Ni atom ensembles connected by a phosphorous atom. These ensembles are efficient for hydrogen adsorption and dissociation, while nitrate can readily adsorb between different ensembles. On Ni2P, the density of ensembles is only about 1/6 of that on Ni(111) resulting in a much lower H saturation coverage which facilitates nitrate coadsorption.
Lin Wei, Da-Jiang Liu, B.A. Rosales, James W. Evans, and Javier Vela, Mild and selective hydrogenation of nitrate to ammonia in the absence of noble metals, ACS Catalysis, 2020, 10, 3618-3628
