Scientific Achievement
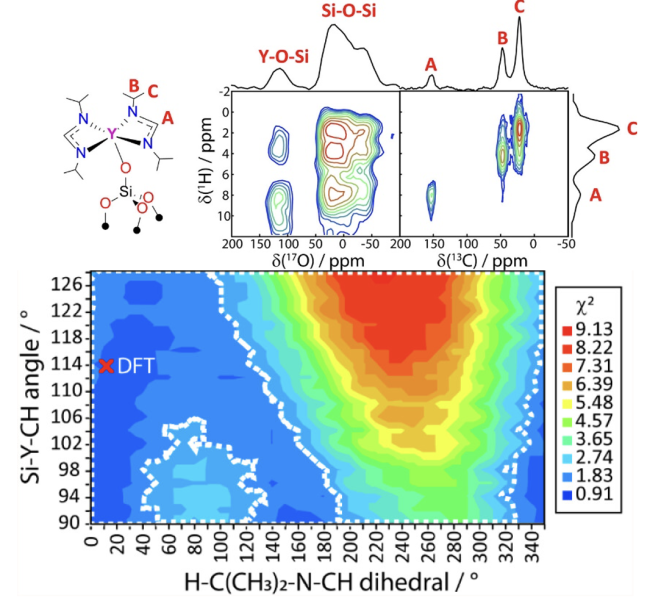
Using dynamic nuclear polarization (DNP)-enhanced 17O NMR we showed that large numbers of quantitative internuclear distances could be determined to effectively triangulate the positions of atoms in a surface site. The new experiment can be easily applied to almost any surface site with minimal synthetic modification.
Significance and Impact
The activity and selectivity of a site in a catalytic reaction is intimately tied to its 3D structure. Determining high-quality and precise structures was previously impossible, which has prevented the formulation of structure-activity relationships.
Research Details
- Protein 3D structures are most often determined using the so-called TEDOR NMR experiment.
- We designed an analogue to the TEDOR experiment that enables the measurement of 1H-17O distances.
- Because the metal-bonded oxygen site from the surface is metal-shifted, it enables the measurement of unambiguous intramolecular distances.
- The new approach is far more accessible than prior methods as spin labels do not need to be introduced.
Supported by the Office of Basic Energy Sciences’ Catalysis Science program under Contract No. DE-AC02-07CH11358
Perras, F. A.; Culver, D. B. J. Phys. Chem. C, 2025, 129, 17069. DOI: 10.1021/acs.jpcc.5c04236
